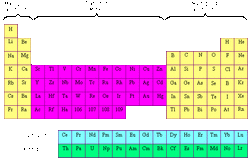
The transition elements are between 2 parts on the periodic table, and 2 line below the big part.
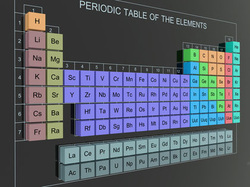
All transitions are metals, so they have the high melt points, great strength, and high density than other metals in the other parts in the periodic table.
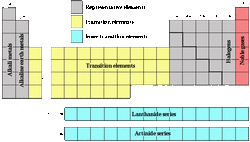
Some transition elements can exist in nature as free elements when it occurs in pure form.

Because of their high melt point great strength, and high density than other metals, so they have many important uses.

Transition elements can react with other elements and form many compounds.
Many of these can be a colorful.
Many of these can be a colorful.

The bottom part’s elements were removed from the main part of the table so that periods 6 and 7 were not longer than other periods.
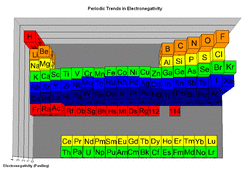
The first row is called lanthanum, would stretch between lanthanum and halfnium. The second row, called the actinium and rutherfordium.
